April 2025
Healthcare facilities worldwide are focusing more on infection control, leading to a surging demand for sterile medical paper packaging. As the number of surgeries, medical treatments, and new drug developments increases, hospitals and pharmaceutical companies need stronger, safer, and more protective packaging. From 2025 to 2034, this market will grow steadily, playing a crucial role in modern medical packaging solutions.
The sterile medical paper packaging market is likely to witness substantial growth during the forecast period. Sterile medical paper packaging is paper-based packaging option that is designed to maintain the sterility of medical devices, surgical instruments, pharmaceuticals and other healthcare products.
These packaging materials comprising medical-grade paper and paperboard, serve as a microbial barrier while allowing sterilization agents like steam, ethylene oxide (EtO), or gamma radiation to penetrate and effectively sterilize the contents. With features such as high breathability, tear resistance as well as reliable sealability, sterile medical paper packaging guarantees product integrity and contamination prevention and makes it essential in hospitals, laboratories and pharmaceutical industries.
The rising prevalence of hospital-acquired infections (HAIs) along with the growing number of surgical procedures worldwide is expected to augment the growth of the sterile medical paper packaging market during the forecast period. Furthermore, the stringent regulatory requirements set by organizations like the FDA and ISO 11607 as well as the shift towards eco-friendly and sustainable packaging are also anticipated to augment the growth of the market.
Additionally, the advancements in sterilization technologies such as ethylene oxide, gamma radiation and electron beam sterilization as well as the expansion of the pharmaceutical and biotechnology industries with the rise in biologics and vaccines is also projected to contribute to the growth of the market in the near future.
The growth in surgical procedures and the increasing utilization of medical devices owing to the rising prevalence of the chronic diseases, aging populations as well as the advancements in the medical technology is anticipated to support the growth of the sterile medical paper packaging market during the estimated timeframe.
According to the World Health Organization (WHO), a projected 313 million surgical procedures are performed yearly and this number expected to rise due to the increasing access to the healthcare in the developing nations. The shift towards minimally invasive surgeries (MIS) is also likely to increase the demand for sterile disposable medical devices such as catheters, syringes, and surgical instruments, all needing sterile packaging to guarantee infection control.
Additionally, the growing popularity of the cosmetic and plastic surgeries has considerably contributed to the demand for sterile medical paper packaging. According to the International Society of Aesthetic Plastic Surgery (ISAPS), About 35 million aesthetic operations were performed in 2023. In 2023, the total number of surgical and non-surgical operations increased by 3.4% to 34.9 million. With almost 2.2 million procedures performed, liposuction remained the most popular surgical operation in 2023. Breast augmentation, abdominoplasty, eyelid surgery and rhinoplasty were the next most prevalent procedures.
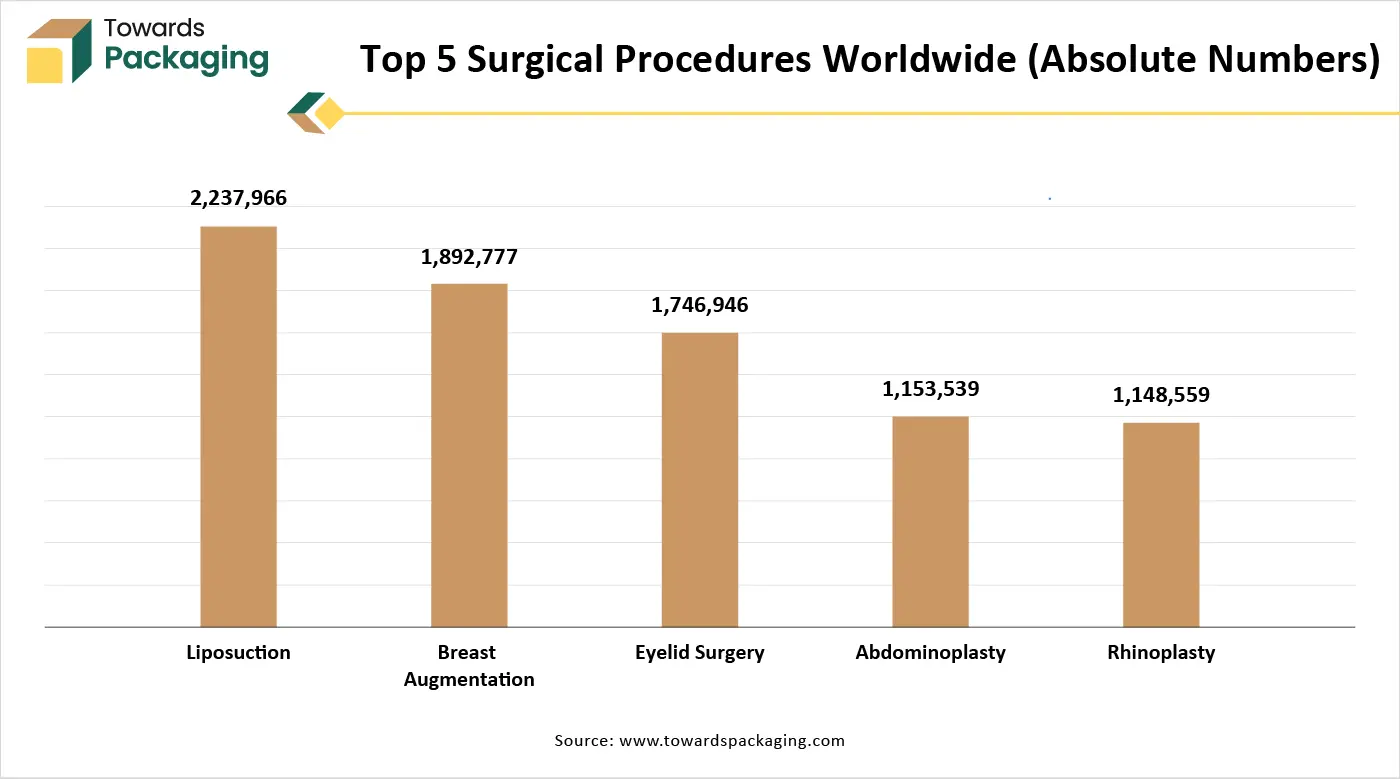
With more than 6.1 million operations, the US led the world in the procedure volume. Brazil came in second with 3.3 million, and first in surgical operations with 2.1 million. These procedures require sterile medical instruments, implants, syringes, and cannulas, all of which must be packaged in contamination-proof, sterile packaging. With the increasing dependence on the surgical as well as the cosmetic procedures along with the technological advancements in the sterilization methods, the sterile medical paper packaging market is expected to witness steady growth within the estimated timeframe.
The stringent regulatory compliance and lengthy approval processes imposed by health and safety authorities are likely to hamper the growth of the sterile medical paper packaging market during the estimated timeframe. Medical packaging must fulfill the strict standards to guarantee the sterility, product integrity and patient safety. Regulatory bodies such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), ISO 11607, and EU Medical Device Regulation (MDR) have set detailed requirements for packaging materials, sterilization validation labeling, as well as for the barrier properties.
If the medical devices need to be sterilized, the packaging needs to be made to keep it that way. The FDA (21 CFR 880.6860) regulates the sterilization procedures and validations. To guarantee that the packaging procedure fulfills its stated purpose and does not risk the medical device's safety and efficacy, it must be validated (as per 21 CFR 820.130).
In the European Union's, the Medical Device Regulation (EU) 2017/745 outlines specifications for the medical devices that involves packaging as well. The MDR highlights how important it is to make certain medical devices are safe and effective for the duration of their lives. The EU, like the FDA, mandates adherence to ISO 10993-1: XXXX for the biological assessment of materials utilized in medical device packaging.
Moreover, compliance with these regulations mostly leads to prolonged approval timelines, delaying the product launches as well as increasing the costs for manufacturers. Additionally, companies face challenges in adapting to the changing global standards, which further complicates the market entry. Thus, the complexity and stringent approval processes create barriers for new entrants and slow down innovation in sterile medical paper packaging.
The expansion of the healthcare infrastructure and rising investments in the hospital facilities to manage patient care efficiently attributed to the increasing prevalence of chronic diseases such as diabetes, cardiovascular disorders and cancer is expected to create immense opportunities for the growth of the sterile medical paper packaging market in the years to come. Many economies are witnessing rapid growth in the healthcare infrastructure with investments in opening of new hospitals.
For instance:
Furthermore, governments across the globe are also investing in healthcare infrastructure. For instance, the Saudi Arabian government intends to invest more than $65 billion under Vision 2030 to build the nation's healthcare infrastructure, restructure and commercialize insurance and health services, establish 21 health clusters throughout the nation and increase the availability of e-health services. With governments and private sectors expanding healthcare infrastructure, the demand for the sterile medical paper packaging will continue to rise in the future.
Artificial intelligence is reshaping industries worldwide and the sterile medical paper packaging market is no exception. As the technology keeps on advancing, AI based innovations are set to transform sterile barrier packaging, improving the efficiency, precision as well as compliance while introducing new challenges. From automating quality control to optimizing the sustainable packaging options, AI is redefining the way medical packaging guarantees sterility and reliability in the healthcare sector. Sterile barrier packaging can be carefully inspected for the flaws with AI systems that can also provide strict quality control. Manufacturers can detect every minor defect through the utilization of computer vision and machine learning techniques, which lowers the possibility of sterility being compromised. This leads in safe products as well as more confidence from the customers.
Furthermore, AI automation can improve the productivity with streamlining packing processes. Equipment failures can be predicted utilizing the predictive maintenance models that reduces downtime and improves the production schedules. As a result, turnaround times are shortened, expenses are decreased, and total efficiency is increased.
Additionally, AI makes it possible to create customized packaging ideas to fulfill certain needs. Manufacturers can utilize AI tech to develop packaging designs that appeal to the specific groups through examining data on the customer preferences. This degree of personalization can increase customer engagement and usage while strengthening brand loyalty. With continuous advancements in the AI technology, quality assurance as well as sustainable packaging, the sterile medical paper packaging market is expected to experience greater efficiency, shaping the future of the sterile packaging in the healthcare industry.
The pouches & bags segment held considerable market share in the year 2024. Paper pouches are an affordable packaging option that can stand exposure to different sterilizing techniques while preserving product sterility until it is time to utilize it. This flexible simple option is well-suited used for packaging low-profile medical instruments, surgical tools, pharmaceutical products, wound care items and diagnostic kits, guaranteeing contamination-free storage and transport. One of the key advantages of the pouches and bags is their lightweight yet durable design and this makes them ideal for single-use medical devices that need aseptic handling. Additionally, pouches and bags also provide customizable sizes as well as barrier properties which are expected to support the growth of the segment during the forecast period.
The pharmaceutical & biological segment held largest share in the year 2024. This is due to the rapid expansion of the biopharmaceutical industry coupled with a surge in drug production and development worldwide. Furthermore, the continued impact of pandemic preparedness programs and global vaccination campaigns as well as the increasing production of mRNA vaccines, monoclonal antibodies and biosimilars is also likely to support the growth of the segment in the global market.
As per the biosimilar report by Samsung Bioepis, FDA approved 61 biosimilars across 17 biological molecules as of September 2024, with 41 being launched in the U.S. Additionally, the advancements in the cold chain logistics along with the temperature-sensitive drug formulations is also expected to contribute to the segmental growth of the market during the forecast period.
North America held considerable market share in the year 2024. This is due to the highly developed healthcare system with well-established hospitals, research centers and pharmaceutical companies in the region. Additionally, the high number of surgical procedures involving general, orthopedic and cosmetic surgeries coupled with the rising focus on the hospital-acquired infection (HAI) prevention is also further expected to contribute to the regional growth of the market.
According to the statistics by the American Society of Plastic Surgeons (ASPS), in 2023, liposuction continued to be the most popular plastic surgery operation, with 347,782 procedures carried out, a 7% rise from 2022. With 170,110 procedures conducted, tummy tucks also witnessed a 5% increase, indicating continued demand in these life-changing treatments. Also, the rise of the home-based healthcare services as well as the remote patient monitoring are also expected to support the regional growth of the market.
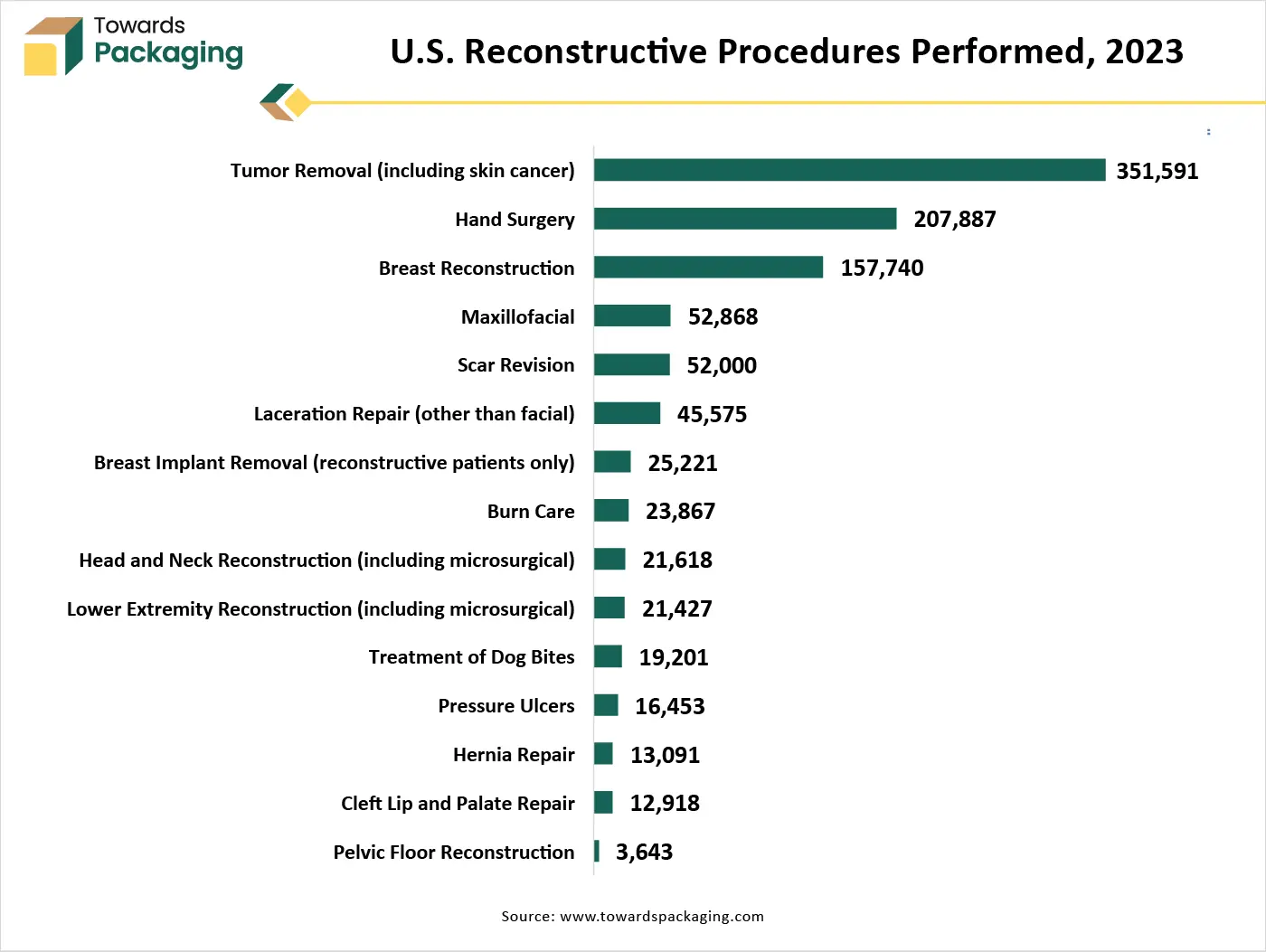
Asia Pacific is likely to grow at a fastest CAGR during the forecast period. This is due to the shift toward single-use medical instruments, syringes and surgical kits to reduce cross-contamination risks. Furthermore, the expansion of medical tourism industry is also expected to contribute to the regional growth of the market.

By Product Type
By Sterilization Method
By Application
By Region
April 2025
April 2025
April 2025
April 2025